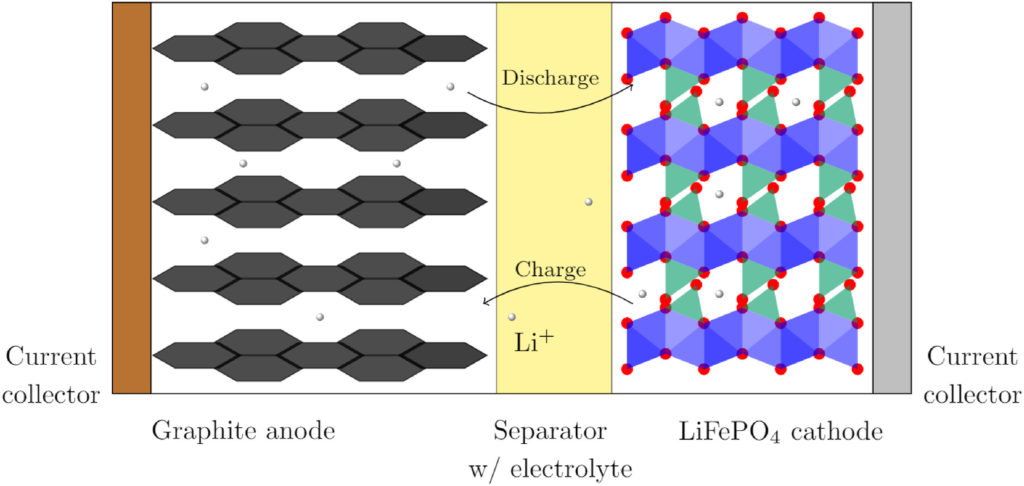
Lithium ion batteries are used to store and produce electric energy. During the charging- and discharging processes, net heat is released or adsorbed in the battery. Temperature is one of the most important factors for battery ageing. In this study we are seeking a fundamental understanding of one of the heat effects, the reversible heat source. The role played by local reversible heat changes is mostly overlooked. We show that, despite a small full cell battery entropy change, there are large reversible half cell heat effects of opposite signs in the lithium iron phosphate oxide and lithium graphite electrode compartments. We present for the first time the Peltier heat of the LiFePO4 electrode near 0% state of charge. The results are documented by thermoelectric potentials which are measured over days to achieve a stationary state. Experiments, supported by theory, enable us to decompose two processes with different characteristic times. We report effective diffusion coefficients for LiPF6, and diethyl carbonate.
Read the full article from Gunnarshaug et al. here: https://doi.org/10.1016/j.electacta.2019.135567